Absolute entropy of water calculator4/14/2024 There is no difference in calculational technique from Example #1. This is the typical situation in this type of problem.Įxample #2: Determine the final temperature when 10.0 g of aluminum at 130.0 ☌ mixes with 200.0 grams of water at 25.0 ☌. Note that the iron drops quite a bit in temperature, while the water moves only a very few (2.25 in this case) degrees. If you aren't too fussy about significant figures. Please note the use of the specific heat value for iron. Solution Key Number Two: the energy amount going out of the warm water is equal to the energy amount going into the cool water. To compute the absolute distance, it's the larger value minus the smaller value, so 85.0 to x is 85.0 minus x and the distance from x to 20.0 is x minus 20.0 That last paragraph may be a bit confusing, so let's compare it to a number line: The colder water goes up in temperature, so its Δt equals x minus 20.0. The warmer iron goes down from to 85.0 to x, so this means its Δt equals 85.0 minus x. Solution Key Number One: We start by calling the final, ending temperature 'x.' Keep in mind that BOTH the iron and the water will wind up at the temperature we are calling 'x.' Also, make sure you understand that the 'x' we are using IS NOT the Δt, but the FINAL temperature. This is very, very important.Ĭ) The energy which "flowed" out (of the warmer water) equals the energy which "flowed" in (to the colder water) The warmer metal will cool down (heat energy "flows" out of it).ī) The whole mixture will wind up at the SAME temperature. Forgive me if the points seem obvious:Ī) The colder water will warm up (heat energy "flows" in to it). These values are tabulated and lists of selected values are in most textbooks.Įxample #1: Determine the final temperature when a 25.0 g piece of iron at 85.0 ☌ is placed into 75.0 grams of water at 20.0 ☌.įirst some discussion, then the solution. 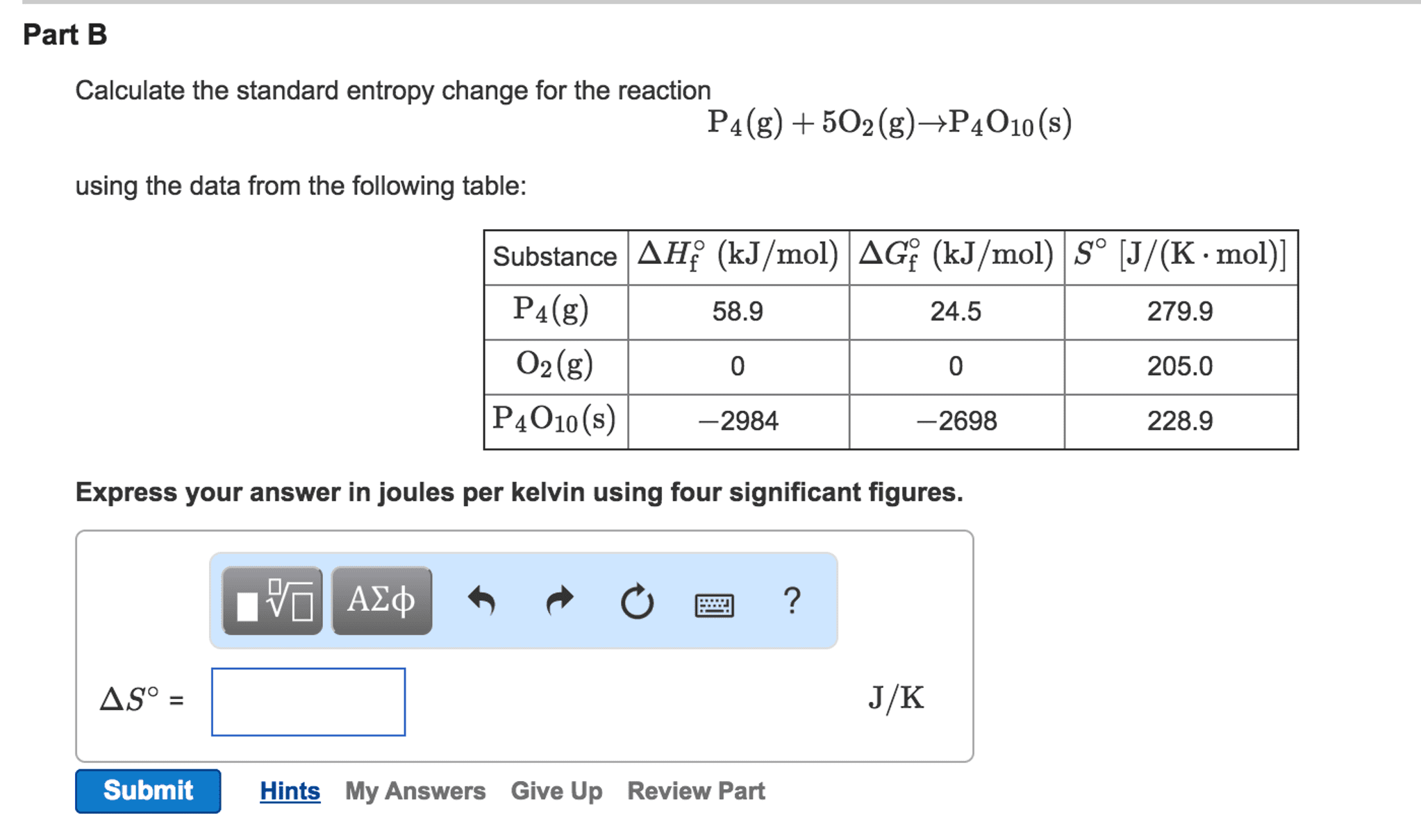
The water specific heat will remain at 4.184, but the value for the metal will be different. These problems are exactly like mixing two amounts of water, with one small exception: the specific heat values on the two sides of the equation will be different. Go to calculating final temperature when mixing two samples of water Go to calculating final temperature when mixing metal and water: problems 1 - 15 Note that the Excel program uses SI units for the thermodynamic property values.The final temp after warm metal is put into colder water When warm metal is put into colder water, what final temperature results? The program is compatible with all versions of Excel.Ĭlick on this link to see a screen capture of the Excel spreadsheet for the superheated region. 
To use the calculator you need to have Microsoft Excel installed on your computer. You need to uncompress this file before you can access the calculator. In the subcooled and superheated region, you can specify ( P, T), ( P, v), ( P, u), ( P, h), or ( P, s), and automatically solve for the remaining thermodynamic properties.Īll the files that come with this calculator are contained in a single (compressed) file, in the "zip" format. In the saturated region, you can specify any of the six thermodynamic properties and automatically solve for the remaining five. 
It calculates thermodynamic properties in the subcooled region for an absolute pressure range of 5-50 MPa, using 7 data tables spread apart over this range. The Excel spreadsheet calculates thermodynamic properties in the superheated region for an absolute pressure range of 0.01-80 MPa, using 30 data tables spread apart over this pressure range. The calculator uses data points from thermodynamic property tables, and uses linear interpolation to calculate the following six thermodynamic properties: Temperature ( T), pressure ( P), specific volume ( v), energy ( u), enthalpy ( h), and entropy ( s). This calculator is programmed in Microsoft Excel. I created a steam table calculator which makes it easy to calculate the thermodynamic properties of water in the subcooled, saturated, and superheated region.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
